EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L
(税込) 送料込み
商品の説明
商品情報
【商品名】
11581円EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L家具、インテリアインテリア雑貨EKO ゴミ箱 EK9233MT 20L+20L ブラヴィア ダストボックス 自動感知 オートセンサービン 分別 ステンレス製 ごみ箱 ふた付き 便利 自動開閉 横型 スリム
EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L
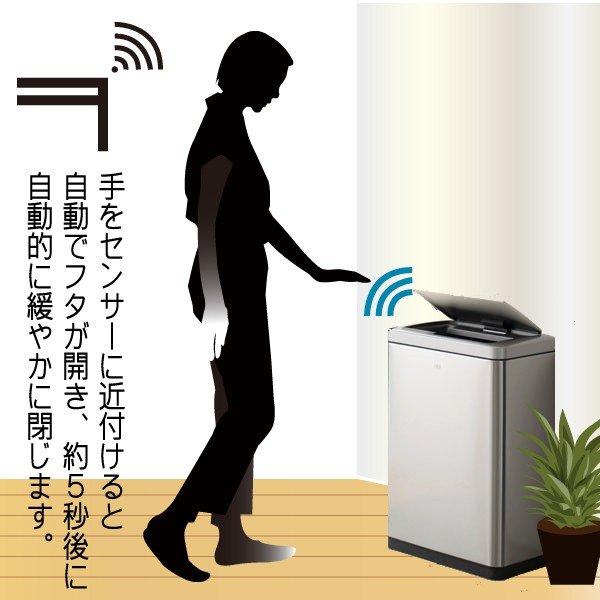
【商品説明】
・サイズ:W46×D27×H64.7cm、フタ開口時H85.5cm
【サイズ】
高さ : 33.40 cm
横幅 : 53.00 cm
奥行 : 74.00 cm
重量 : 10.84 kg
※梱包時のサイズとなります。商品自体のサイズではございませんのでご注意ください。EKO ブラヴィアセンサービン 20+20L EK9233MT-20L+20L (ゴミ箱(ごみ箱
EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L - ゴミ箱
EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L
EKO ブラヴィアセンサービン 20+20L EK9233MT-20L+20L (ゴミ箱(ごみ箱
EKO ゴミ箱 EK9233MT 20L+20L ブラヴィア ダストボックス 自動感知 オートセンサービン 分別 ステンレス製 ごみ箱 ふた付き 便利 自動開閉 横型 スリム
EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L - ゴミ箱
EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L
EKO ブラヴィアセンサービン 20+20L EK9233MT-20L+20L (ゴミ箱(ごみ箱
Amazon.co.jp: EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L
EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L - ゴミ箱
楽天市場】EKO ゴミ箱 EK9233MT 20L+20L ブラヴィア 自動開閉 自動感知
旧商品 【国内正規輸入品】 EKO ゴミ箱 ブラヴィア センサービン 20L+20L ステンレス 分別 EK9233MT-20L20L 送料無料
EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L - ゴミ箱
EKO ブラヴィアセンサービン 20+20L EK9233MT-20L+20L (ゴミ箱(ごみ箱
楽天市場】EKO ゴミ箱 EK9233MT 20L+20L ブラヴィア 自動開閉 自動感知
EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L - ゴミ箱
EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L - ゴミ箱
EKO ブラヴィアセンサービン 45L EK9233MT-45L (ゴミ箱(ごみ箱)) 価格
EKO ブラヴィア センサービン EK9233MT-20L+20L単3乾電池6本使用 - ごみ箱
楽天市場】EKO ゴミ箱 EK9233MT 20L+20L ブラヴィア 自動開閉 自動感知
EKO ゴミ箱 EK9233MT 20L+20L ブラヴィア ダストボックス 自動感知 オートセンサービン 分別 ステンレス製 ごみ箱 ふた付き 便利 自動開閉 横型 スリム
EKO ブラヴィアセンサービン 20+20L EK9233MT-20L+20L (ゴミ箱(ごみ箱
EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L
楽天市場】EKO ゴミ箱 EK9233MT 20L+20L ブラヴィア 自動開閉 自動感知
Amazon|ゴミ箱 ごみ箱 ダストボックス 自動開閉式 2分別 20L+20L 20
楽天市場】EKO ゴミ箱 EK9233MT 20L+20L ブラヴィア 自動開閉 自動感知
EKO ゴミ箱 EK9233MT 20L+20L ブラヴィア ダストボックス 自動感知 オートセンサービン 分別 ステンレス製 ごみ箱 ふた付き 便利 自動開閉 横型 スリム
EKO ブラヴィア センサービン EK9233MT-20L+20L単3乾電池6本使用 - ごみ箱
Amazon.co.jp: EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L
EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L - ゴミ箱
Amazon.co.jp: EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L
EKO ゴミ箱 EK9233MT 20L+20L ブラヴィア ダストボックス 自動感知 オートセンサービン 分別 ステンレス製 ごみ箱 ふた付き 便利 自動開閉 横型 スリム
日用品 掃除 関連商品 ステンレス製ゴミ箱(ダストボックス) ブラヴィア
EKO ブラヴィア センサービン EK9233MT-20L+20L単3乾電池6本使用 - ごみ箱
サンコウ EK9233MT 20L+20L ダストBOX 4531833365180 1台(直送品
EKO ブラヴィア センサービン 20L+20L EK9233MT-20L+20L
EKO ブラヴィア センサービン EK9233MT-20L 20L editorial.unju.edu.ar
日用品 掃除 関連商品 ステンレス製ゴミ箱(ダストボックス) ブラヴィア
楽天市場】【12/1限定!全品3%OFF&最大P37倍※ワンダフル+SPU+独自P
日用品 掃除 関連商品 ステンレス製ゴミ箱(ダストボックス) ブラヴィア
商品の情報
メルカリ安心への取り組み
お金は事務局に支払われ、評価後に振り込まれます
出品者
スピード発送
この出品者は平均24時間以内に発送しています